Checkmate 9la study design9/7/2023  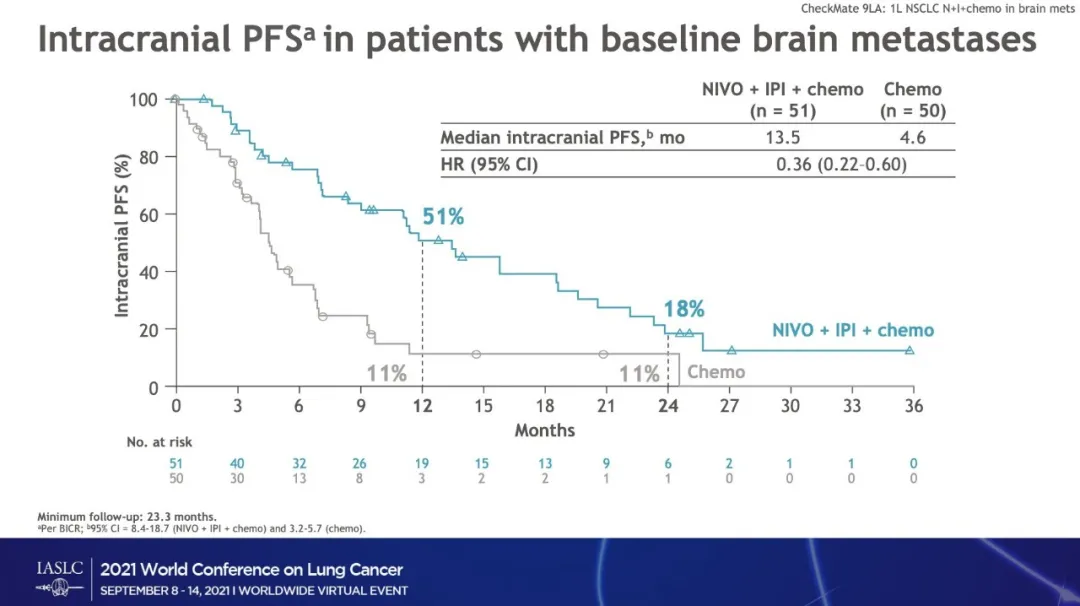
Have that option of giving the 2 cycles of chemotherapy first with the immunotherapy to make sure there isn’t early progression. This will give clinicians an option when the patient has a significant tumor volume and they’re symptomatic from their cancer, and the clinician is concerned that if the initial treatment doesn’t control the cancer, the patient will get into trouble and then we won’t be able to get additional treatment. Many patients want to avoid chemotherapy when they can or minimize exposure to chemotherapy. The true measure for all these regimens will really be long-term survival and which approach leads to more durable survival at landmark rates. Defining a specific role for all patients is challenging, and in the right clinical context it certainly can provide value. It has to be for the right patient in the right clinical circumstance. The rates of anemia and neutropenia were a bit lower with the 9LA regimen, reflecting less cumulative toxicity from chemotherapy, perhaps less long-term neuropathy. The rates of grade 3/4 adverse events and the discontinuation rates were higher certainly with the CheckMate 9LA regimen compared with chemotherapy alone, but some of the toxicity rates were better. There is certainly more toxicity when we’re adding treatments to it. When we’re adding chemotherapy to dual-checkpoint blockade, there will be a cost. By preventing continued chemotherapy, will we avoid more cumulative toxicity? Will we enable more development of memory T cells by preventing the potentially detrimental effect of cytotoxic chemotherapy over time? It’s an interesting strategy limiting chemotherapy to those first 2 cycles. When you think you may only have 1 opportunity to deliver treatment for those patients, I think including cytotoxic chemotherapy is of value. In someone with aggressive disease, a high burden of disease, that initial use of chemotherapy is appealing to prevent that early drop-off, that early progressive event. Stephen Liu, MD: The CheckMate 9LA regimen giving 2 cycles of histology-specific chemotherapy with dual-checkpoint blockade is another option, and we have to tailor the treatment options to the specific patients we see. 
It is useful for the different studies to make sure you’re looking at the outcome for the specific histologies, because that can be helpful in guiding appropriate choice of treatment. They have slightly different eligibility criteria, but it’s only natural that when we’re choosing treatment for patients we’re trying to get the most effective treatment. We shouldn’t be carefully comparing the different trials because they’re not exactly in the same population or in the same countries. A better outcome for the nonsquamous compared with the squamous. In the CheckMate 227 study, nivo-ipi had a median survival of over 19 months for the nonsquamous group, and in CheckMate 9LA survival was over 17 months. If we look at histology-based outcomes like 227 and 9LA, we see better survivals in the nonsquamous group. Keep in mind that for CheckMate 227 and 9LA, what we see is a composite. We have data for chemotherapy–I/O in nonsquamous lung cancer and chemotherapy–I/O in squamous cell lung cancer, and overall survival tends to be better in the nonsquamous setting compared with the squamous setting. The CheckMate 227 trial and the CheckMate 9LA trial were for all histologies, whereas the other randomized trials in this setting-for instance, KEYNOTE-189, KEYNOTE-407, IMpower150, IMpower130-were histology specific. Ready, MD: It’s natural for us to compare trials. But the overall approach of giving 2 cycles, a limited course of histology-specific chemotherapy with dual-checkpoint blockade, should apply across histologies. 
This is a regimen where histology will dictate which chemotherapy you deliver with those first 2 cycles. When we break it down by histology, we did see a consistent benefit, a survival benefit in both the squamous and nonsquamous histologies with fairly comparable outcomes and comparable improvements. Factoring in those numbers, the outcomes are really impressive. We know from multiple randomized trials, including some cross-trial comparisons, that our outcomes in the squamous histology are a bit worse. About 30% of patients were squamous, which is a bit higher than some of the other studies. Stephen Liu, MD: When we look at CheckMate 9LA, this trial did include both squamous and nonsquamous histology. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
